 
Plasma formation is initiated by a spark from a Tesla coil. The sample is mixed with a stream of Ar using a nebulizer, and is carried to the plasma through the torch’s central capillary tube. The ICP torch consists of three concentric quartz tubes, surrounded at the top by a radio-frequency induction coil. Because a plasma operates at a much higher temperature than a flame, it provides for a better atomization efficiency and a higher population of excited states.Ī schematic diagram of the inductively coupled plasma source (ICP) is shown in Figure 10.7.2 A plasma’s high temperature results from resistive heating as the electrons and argon ions move through the gas. The plasma used in atomic emission is formed by ionizing a flowing stream of argon gas, producing argon ions and electrons. We also expect emission intensity to increase with temperature.Ī plasma is a hot, partially ionized gas that contains an abundant concentration of cations and electrons. Essentially, the spectrum that we observe is the continuous spectrum with dark spots where the gas has "picked out" the continuum.We expect that excited states with lower energies have larger populations and more intense emission lines. The above examples have been of emission spectra however, there is another type called "absorption spectra." Sometimes when we view sunlight through a gas, we get a spectrum which is continuous except for the regions in which the gaseous element would have emitted its signature spectra if heated. This type of spectrum shows us not only where the Sun emits light, but also gives a measure of how much light is emitted as a function of energy. Notice that where the first spectrum shows a bright line, the second shows a peak in the graph. Compare the two representations of the solar UV spectrum. While the graph isn't as colorful as those shown in a "photographic" format, this representation tells us much about the UV emission of the Sun. However, instead of indicating the observed energies with light bands, the graph shows the intensity of the radiation on the y-axis. As with the other spectra, the x-axis indicates the energy of the observed light. The graph below the Sun's UV spectrum shows the same information in a more quantitative format. There is no quantitative measure of the light emitted as a function of energy. In that representation, we can see where the emission by the Sun is most active, but again we see little about the intensity of the radiation. The spectrum covers wavelengths of energy between 300 and 350 angstroms. Remember that our eyes can't see ultraviolet light however, as with the visible spectra shown above, the horizontal axis (the x-axis) shows the energy (or "color") of the observed light. 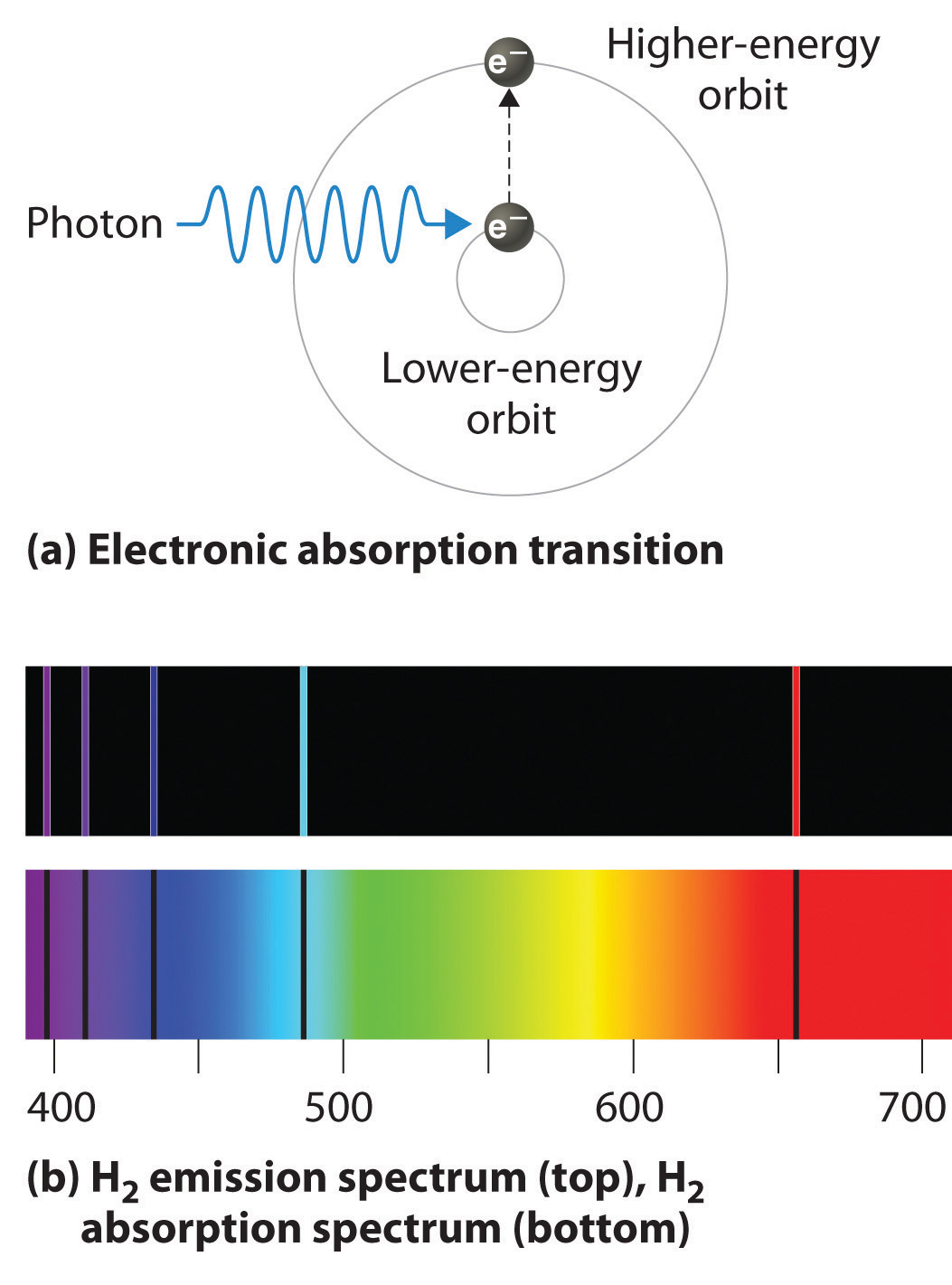
This representation has been modeled after the visible spectra. The top spectrum shows a narrow segment of Sun's spectrum of ultraviolet (UV) light. The bottom shows a graph of the same spectrum. The top shows an image of a portion of the solar ultraviolet spectrum. Two different representations of the same spectrum. Scientists, then, tend to use a different representation which permits a more rigorous analysis. On top of that, they would convey no information at all if it the observed radiation was outside the limits of the human eye. We can't really see how much light there is of each of the colors they seem about equally bright, and it would be hard to make a quantitative comparison. While the colors in the above representations of spectra are pretty, their usefulness is limited. These type of spectra can be generated for many elements by vaporizing the element in a flame. The one shown above characteristic of carbon. It turns out that each chemical element emits its own characteristic pattern. In the image above, the horizontal axis shows the energy (or color) of the light, the same as the spectrum above, the light is emitted only at particular "discrete" energies corresponding to the bright lines. Clearly, this is not a continuous spectrum however, it still represents visible radiation that has been separated into its constituent colors. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
